Cardiac Science Powerheart AED G5
Cardiac Science Powerheart AED G5
Couldn't load pickup availability
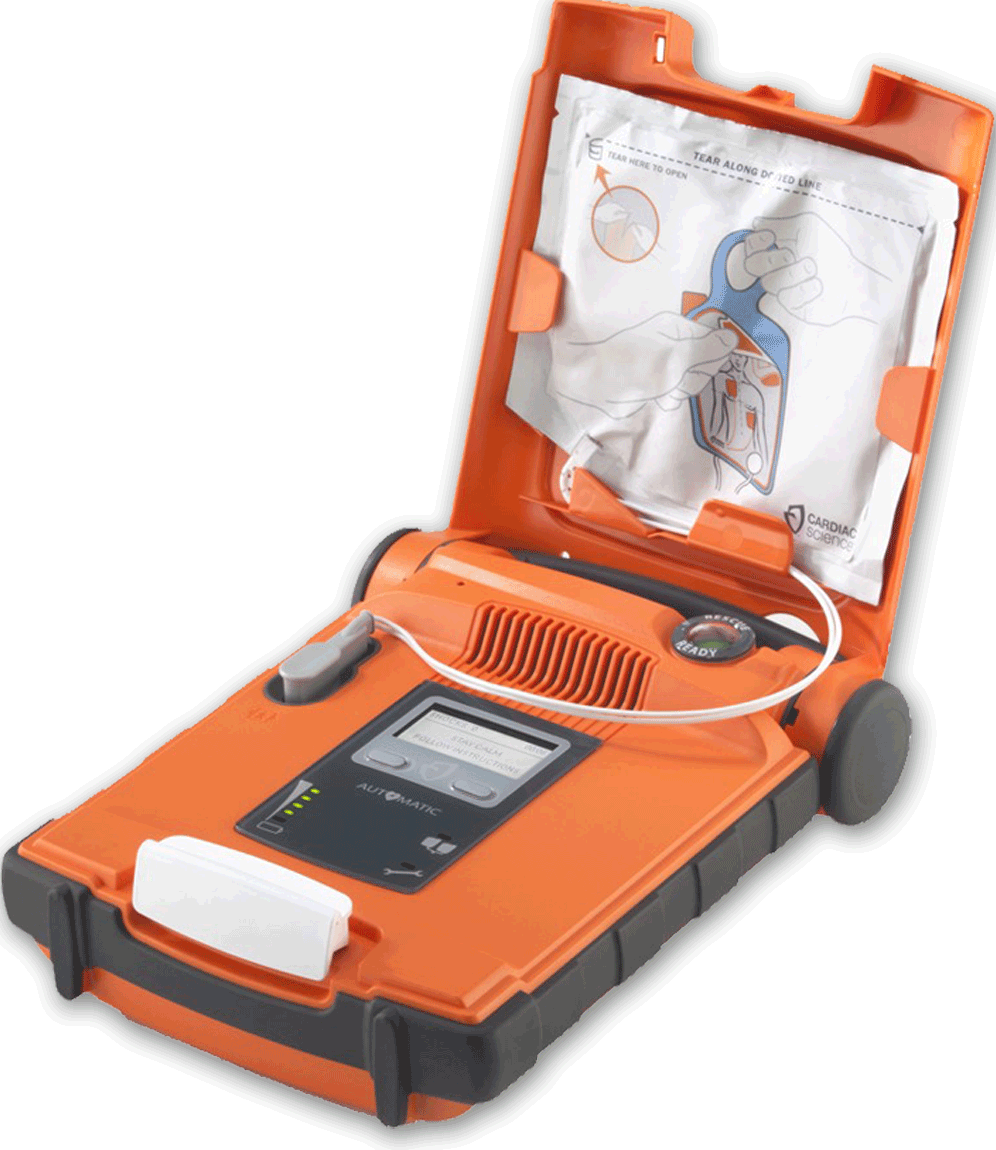
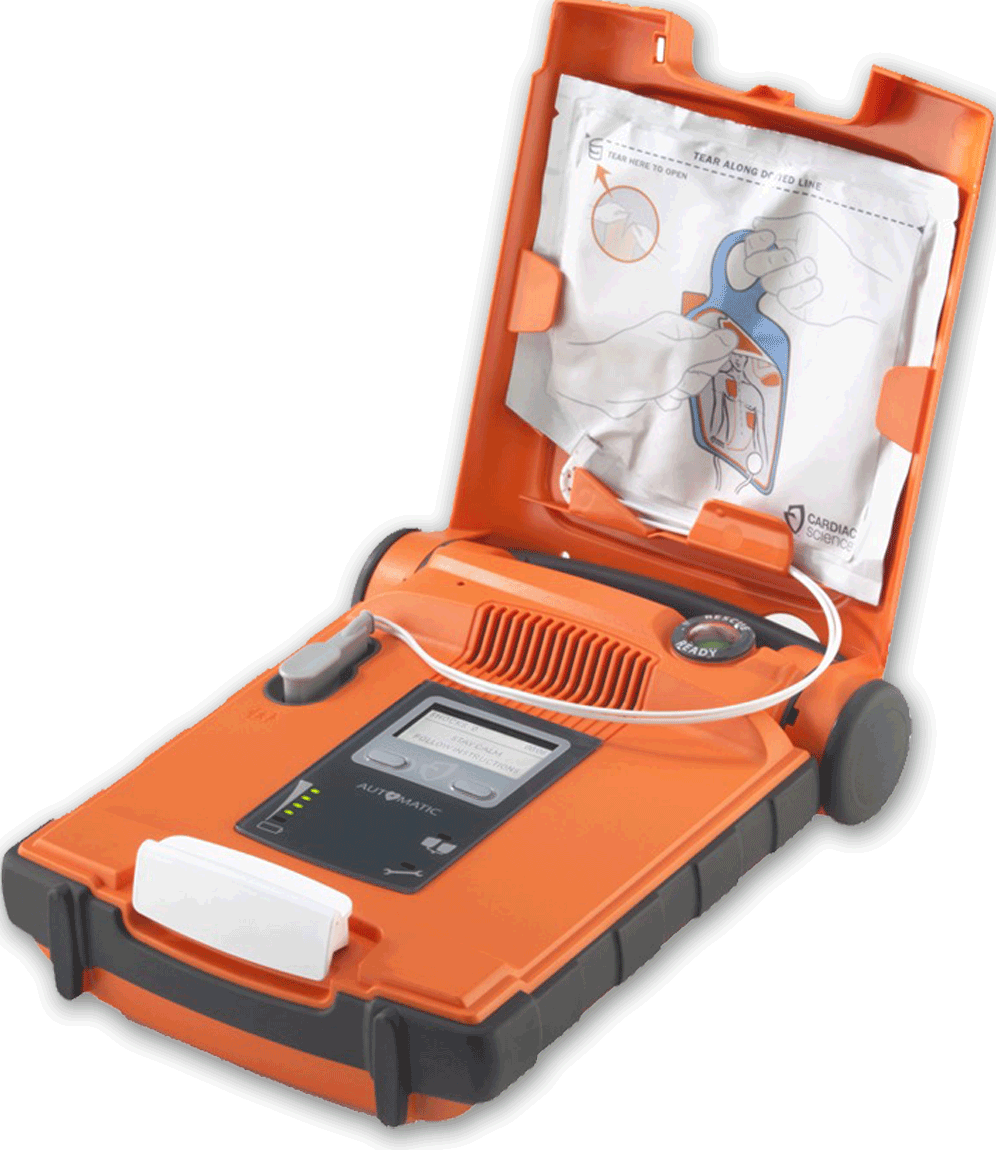
In 2015, Cardiac Science was granted FDA approval to introduce the fully-automatic Powerheart® G5 AED in the United States, and in Canada in July 2017. It is the first FDA-approved AED combining fully automatic shock delivery, dual-language (English to Spanish/French) option, variable escalating energy and fast shock times. Readers/users with an understanding of the AED revolution/evolution will appreciate the combination of technology and features of this G5 AED and the niche that it fills. Trained or untrained; novice or professional – anyone who arrives first at the scene of a sudden cardiac arrest event can begin life-saving guided therapy with this G5 AED that may well save a life.
What's inside:
- Powerheart® AED G5 with 8-Year Warranty
- Two Sets of Standard or One Set of Intellisense® CPR Adult Electrode Pads
- Intellisense® Battery Pack with 4-Year Warranty
- User Guide, Steps to Rescue, Getting Started Software
- Premium Carrying Case
- USB Data Cable
- AED Manager Software
- Premium AED/CPR Responder® Pack
- “AED Equipped” Window/Wall Decal
- AED Inspection Tag
- Physician’s Prescription
The G5 AED is easy to use, packed with advanced technology and is high-performance in harsh settings, even meeting the military’s field standards. The Rescue Ready® technology runs daily, weekly and monthly self-checks on the AED. The RescueCoach™ guides and paces the rescue in 3 languages (English Spanish and French) while the optional Intellisense® Defibrillator Pads (iCPR pads) contain a CPR-smart sensor that measures and provides feedback on effective CPR delivery. Standard adult and pediatric pads are also available. The Cardiac Science G5 AED is the culmination and next generation of over 100 patents and 20 years of engineering. They have filled a need for an AED that is both user-friendly and packed with life-saving technology.
Physical Description and Characteristics:
- (H X W X D) 3.4” X 9.0” X 11.8”
- Weight 5.7 lbs
Maintenance and Management for your Cardiac Science Powerheart® G5 AED:
All AEDs require regular maintenance and management to ensure that the AED will perform during a sudden cardiac arrest (SCA). The electrodes and batteries must be tracked for expiration dates and replacement. Your organization should have one person in charge of this very important task and be able to access records demonstrating maintenance and oversight.